

A pulseometer is a wearable device to measure a user's real-time heart rate. Pulseometers are commonplace for use in both medical settings and at-home patient care. Commercial pulseometers measure heart rate (bpm) and peripheral blood oxygen saturation (SpO2). These measurements are useful for everyday health monitoring, but are particularly important for patients with conditions affecting the lungs or heart - like COPD, COVID-19, or heart disease.
This Preliminary design report will give the underlying theory for this device and outline our plan for the design of this device, with the goal that it could be marketed as a commercially viable finger clip pulseometer. It will incorporate real-time heart rate monitoring with a user-friendly design and visual display. Using the principles of photoplethysmography, we will be able to detect the subtle flushing that occurs when blood flows through capillary arteries near the surface of the skin. Then, using various signal processing methods, we will turn this information into a heart rate. This device will not measure oxygen saturation as that is outside the scope of this course, but could be considered in future iterations of this project.
Our pulseometer, like commercial products, is based on photoplethysmography (PPG). PPG is an optical technique based on the Beer-Lambert law, and it can be used to measure physiologic information about a patient. When a patient’s skin is illuminated with specific wavelengths of light, the absorbance fluctuates in accordance with the flow of blood through that area of tissue. If one can measure this real-time absorbance, it is possible to extract information about not only heart rate, but blood oxygen saturation, respiratory rate, and even sleep quality
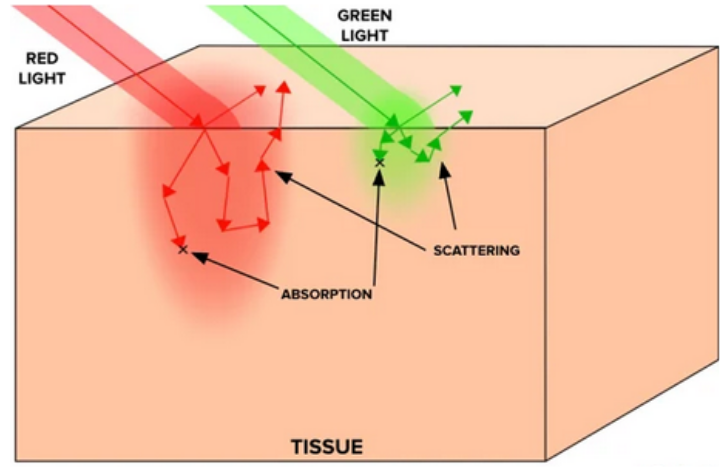
Our project will use transmittance PPG, where the light source is positioned opposite to the light collecting device, so the signal must travel through the appendage (fingertip) as demonstrated in the figure. In order to pick up small absorbance changes in pulsatile artery blood, we need to understand the optical properties of tissue and blood. Considering we are using transmittance PPG, light must be able to travel through the fingertip without getting completely absorbed or scattered out. Light scattering in tissue can be modeled with a power law relationship:
Depending on the tissue, b could be anywhere between 0.7-1.5. It is clear that as wavelength decreases, scattering increases, and we see a similar trend for absorption. Absorption follows the Beer-Lambert law:
Which can be re-expressed as:
Re-arranging the expressions above we see that μa ∝ cε. However, in tissue samples this becomes slightly more complicated, as the Beer-Lambert law assumes the presence of only one absorber. In skin and tissue there are many absorbers, so we can express this with a sum:
Each of these absorbers has slightly different molar extinction coefficients (melanin and other chromophores in the skin exhibit strong light absorption in the 200-500nm range, whereas water and lipids become more significant past 800nm). Absorbance is not as simple as scattering, but in general molar extinction coefficients for relevant organic molecules in the skin become stronger at lower wavelengths. Therefore if the goal is for light to travel all the way through tissue, it is more helpful to use a higher wavelength of light. It must still be a wavelength that can pick up on vascular activity; that is, it must be absorbed or scattered just enough so that we can detect a change when blood flow is occurring.
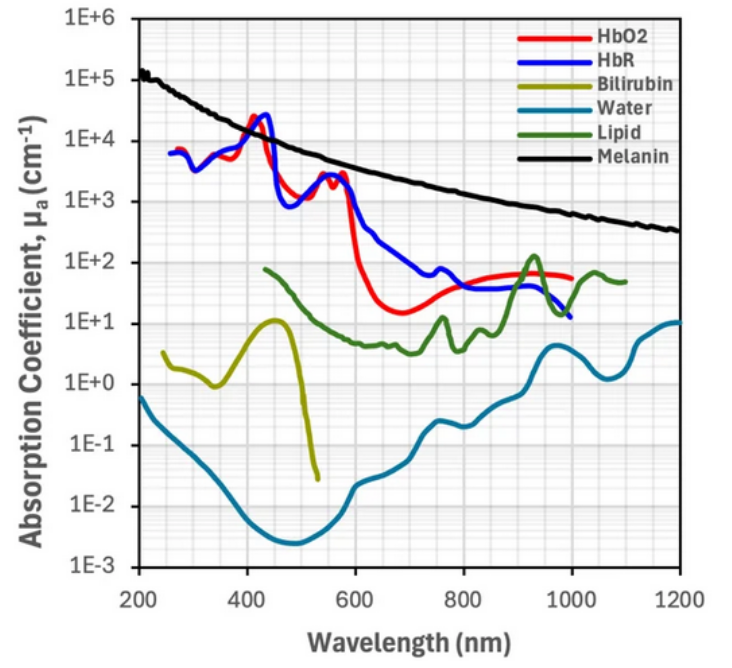
Deoxygenated and oxygenated hemoglobin absorb some wavelengths light equally (red-NIR), but some wavelengths differently, and this contrast allows us to collect absorbance data. Infrared light (850-1000nm) is more absorbed by oxygenated hemoglobin, while deoxygenated hemoglobin has stronger absorption of red light (650–750nm). In commercial devices, the difference between these absorbance ranges allows one to measure blood oxygen saturation. However, for the purpose of this device we will therefore be using infrared light, as it will have enough penetration depth into the tissue to be detected with transmittance PPG while also being sufficiently scattered / absorbed by oxygenated blood for us to detect it.
Challenges in producing an effective & accurate pulsometer relate to understanding the interaction of light with human skin tissue, small signals with a large amount of background noise, user interaction with the equipment.
The focus point for our device is the finger, yet even the composition of the finger is very complex and tissue properties vary widely from person to person. In our device, light must travel through every skin layer (epidermis, dermis, and hypodermis), then through muscle, fascia, nerve tissue, and other connective tissue. Our device should be able to function regardless of skin color (as melanin content can change refractive behavior of light), fat content, or muscle content. While the patient is using the device, it should still be able to function under differing conditions of fingertip perspiration, slight finger movement, or changing external lighting conditions. We plan to account for these differences by using wavelengths of light that work regardless of skin absorbers (like melanin) and target the absorption spectra of oxygenated blood. We will account for friendly user design by using a finger clip that will block out external light sources and will grip tight enough to not be affected by user movement, but not so tight that it cuts off blood flow.
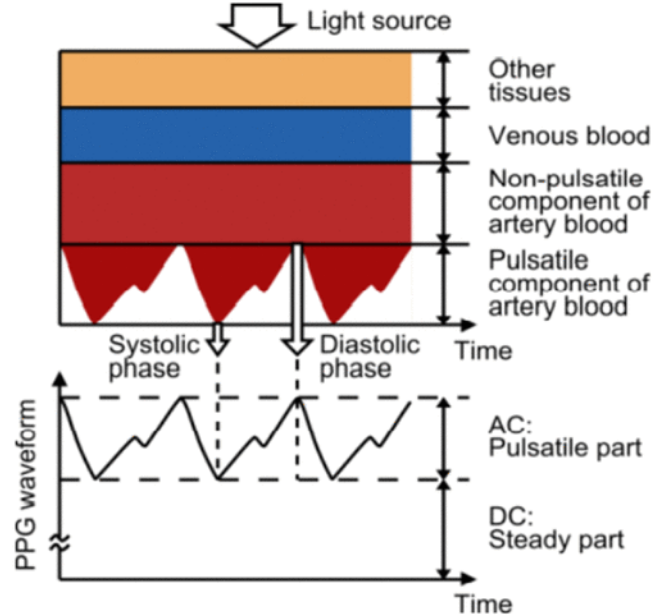
A challenge we faced in CBE 3300A when creating a rudimentary pulsometer is making a small signal amplified and removing background signal and noise. The flushing of the skin that we are trying to detect is very minor, and creating a device to measure these slight changes could be likened to finding a needle in a haystack. There are many sources of background signal and noise for PPG, but absorption from other tissues, background light, and user movement were the largest. However, these factors generally do not change for an individual user while they are using the device, so it tends to result in a baseline DC signal that we have to filter out. What is important is making sure the signal is able to stand out after we remove everything else. We plan to deal with this by implementing new circuit design and signal processing techniques.
Most current circuit design shown below:
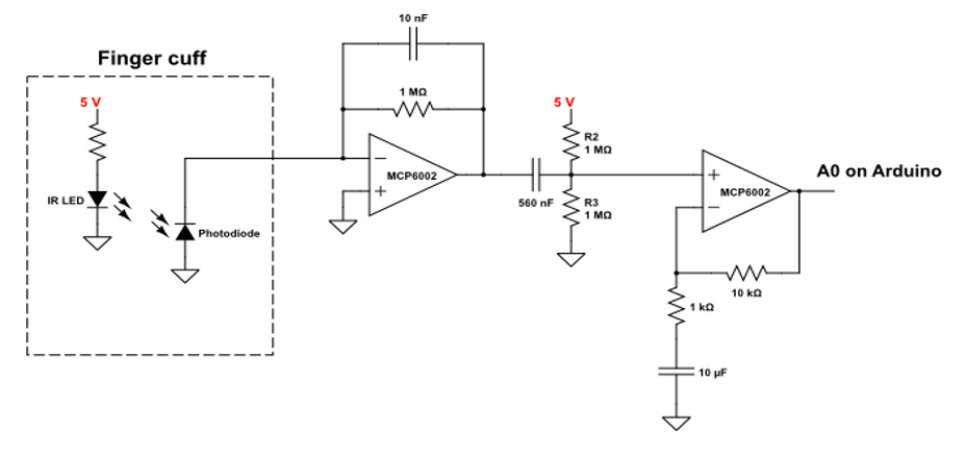
This circuit is made up of four components. Going left to right, the first component is the finger cuff, this is where the patient interacts with the device. The finger cuff will be in the shape of a finger clip, with the IR LED and the photodiode positioned on opposite sides of the finger. The photodiode converts light into electrical current with a high sensitivity and linearity, which is helpful for the small signals we are working with. The second component is the transimpedance amplifier (TIA). The current from the photodiode flows to the negative terminal of the op-amp and through the gain-setting resistor. This creates a voltage at the output of the op-amp that is proportional to the input current - the capacitor helps reduce noise. Next is a simple highpass filter; we are removing the DC baseline signal that comes from the natural absorbance and scattering from the tissue. The last component is a non-inverting gain op-amp. This amplifies the signal by a factor of R2/R1 (the ratio of the resistor in the feedback loop to the resistor connected to ground). The capacitor in this stage blocks the DC signal that we added in the previous stage, amplifying only the AC signal that we care about.
The goal of this whole process is to isolate the signal we care about (absorbance from oxygenated hemoglobin) and amplify it while removing background signal / noise. In this circuit there is already signal processing with multiple built-in filters, but there is more signal processing that has to happen in arduino. In order to remove background noise from the environment, we can use background subtracted signal. We blink the IR LED on and off (50-500Hz) and measure the signal in the on versus off state. Subtracting these two values will give us a reading that is roughly independent of the surrounding conditions - like ambient lighting or different placements of the device on the finger. Furthermore, in order to give a heart rate in bpm, it is necessary to real-time peak detection on the signal. This peak detection becomes very hard with noisy signal, but assuming we can tune our filter appropriately, this peak detection can be done with a simple cut off. If the signal is higher than some predetermined baseline and the signal goes from increasing to decreasing (instantaneous derivative = 0) then we can mark a peak. Time average between peaks gives the users real-time bpm.
The GANTT chart has our projected timeline of the project based on the milestones presented. The deliverables are very direct. There will be six deliverables throughout the project:
The global market for wearable healthcare devices is projected to increase by $75.98 billion by 2030, representing a 67.76% increase over its 2025 value. Over this forecast period, the market is expected to grow at a compound annual growth rate (CAGR) of 10.9%. This shows the demand for wearable healthcare devices will continue to grow. This market includes devices that measure pulse, blood-oxygen levels, glucose, insulin, and more with the purpose of focusing on this market because there isn’t an explicit pulsometer market, only a pulse oximeter market. For the SWOT analysis, we have identified the following:
The goal of this project is to create a commercially viable pulseometer, but in order to do this faithfully we would need to include blood oximetry measurements, as previously mentioned. In order to do this we would need a second LED of a lower wavelength (in the visible red spectrum) and we would need to process that second signal in real time, same as the IR signal. With a device using this dual-LED set up, we could measure far more complex bodily functions, like sleep tracking workout activity, similar to products like a fitbit or apple watch.
Another goal would be to improve the user experience. The circuit described above will be bulky, and therefore difficult to take on the go. If we could miniaturize this circuit on a printed circuit board, and use a much smaller microcontroller, then we could embed the device entirely onto the finger clip, and use bluetooth to communicate with a computer or a phone. Depending on project progress, we would love to try incorporating some or all of these in order to make a more commercially viable product.